Major Points• Types of Radiation (Radio waves, microwaves, infrared, etc.)
• Speed Equation (Speed = wavelength x frequency) • Energy Levels (What happens when an electron becomes energized?) • Pauli Exclusion Principal • Orbital Shapes (S, P, D) • Electron Configuration (Writing out the configuration of the electrons of an element using s, p, and/or d) • Orbital Diagramming (How to diagram the orbitals of an element's electrons using boxes and arrows) • Ionization Energy and Electronegativity in Relation to the Size of an Atom (List the elements from largest to smallest according to their electronegativity, etc.) |
Example Problems1. If the wave speed is 25 m/s and the wavelength is 5, what is the frequency?
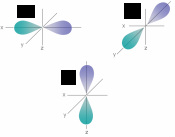
Answer: 5 Hz 2. Identify this orbital: Answer: P orbital
3. What is the electron configuration for Rb? (long form) Answer: 1s^2, 2s^2, 2p^6, 3s^2, 3p^6, 4s^2, 3d^10, 4p^6, 5s^1 4. What is the electron configuration for Rb? (short form) Answer: [Kr] 5s^1 5. What is the electron configuration for F^(-)? Answer: 1s^2, 2s^2, 2p^6 6. Draw the orbital diagram for O. Answer: 7. Draw the orbital diagram for O^-2.
Answer: 8. Draw the orbital diagram for Be^+2
Answer: 9. Which has a higher ionization energy? S or Ca?
Answer: S 10. List these elements in order of electronegativity, from least to greatest: O, Sr, Mg Answer: Sr, Mg, O 11. Which has a greater atomic size? P or Ca? Answer: Ca |
Online Resources
Speed Equation Explanation and Example: http://hyperphysics.phy-astr.gsu.edu/hbase/wavrel.html
Explanation of Orbitals and Identifying Orbitals: http://chemed.chem.wisc.edu/chempaths/GenChem-Textbook/Orbitals-896.html
Atoms and Light Energy: http://imagine.gsfc.nasa.gov/docs/teachers/lessons/xray_spectra/background-atoms.html
Explanation of Orbitals and Identifying Orbitals: http://chemed.chem.wisc.edu/chempaths/GenChem-Textbook/Orbitals-896.html
Atoms and Light Energy: http://imagine.gsfc.nasa.gov/docs/teachers/lessons/xray_spectra/background-atoms.html
Video Resources
Pauli Exclusion Principle and Other Orbital Diagramming Rules: http://www.youtube.com/watch?v=SoNIQjW5Zxs
Electron Configuration (by me!): https://vimeo.com/50656946
Electron Configuration (by me!): https://vimeo.com/50656946
Activity Resources
Chapter 11 Definitions Quizlet: http://quizlet.com/14635148/ch-11-vocabulary-practice-flash-cards/
Electron Configurations Quiz (use periodic table!): http://www.mp-docker.demon.co.uk/as_a2/topics/electronic_configurations/quiz_1.html
Electron Notations Review (use periodic table!): http://www.sciencegeek.net/Chemistry/taters/Unit2ElectronNotations.htm
Electron Configurations Quiz (use periodic table!): http://www.mp-docker.demon.co.uk/as_a2/topics/electronic_configurations/quiz_1.html
Electron Notations Review (use periodic table!): http://www.sciencegeek.net/Chemistry/taters/Unit2ElectronNotations.htm