Major Concepts• Types of Acids and Bases (Arrhenius, Brønsted-Lowry, Lewis)
• Identification (acids, bases, conjugate acids, conjugate bases) • Strong vs. Weak (Acids and Bases) • kw • Finding concentration, pH, and pOH • Determining whether a solution is basic or acidic • Understanding logarithmic functions • Phase Changes • Intramolecular Forces • Intermolecular forces • Sublimation/Deposition • Crystalline Solids |
Example Calculations1. What is the concentration of [OH-] if the [H3O] concentration was 10.0M?
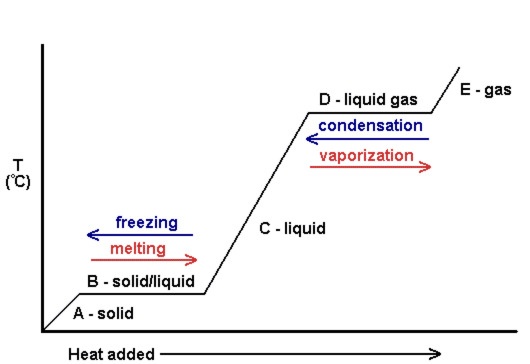
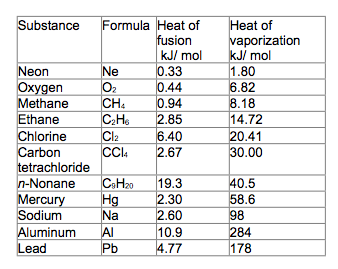
Answer: 1.0x10^-15 M 2. Identify the weak acid: A. H2SO4 B. HCl C. HCH3O D. HNO3 Answer: C. HCH3O 3. If you have 34.5g of methane liquid, calculate the energy you will need to convert the liquid into a gas. Answer: 17.6 kJ 4. If you have 5mol of ice, calculate the energy you will need to turn the ice into water. Answer: 30 kJ 5. Write the conjugate acid for the following base: PO4 (-3 charge) Answer: HPO4 (-2 charge) 6. Write the conjugate base for the following acid: HS (-1 charge) Answer: S (-2 charge) |
Arrhenius Acids & Bases
Acid:
- Any compound that increases H+ (or H3O+) concentration
Example: HCl(g) ----> H+(aq) + Cl- (aq)
HCl is an acid because it increases the H+ concentration
Base:
- Any compound that increases hydroxide (OH-) concentration
Example: NaOH(s) ----> Na+(aq) + OH-(aq)
NaOH is a base because it increases the OH- concentration
- Any compound that increases H+ (or H3O+) concentration
Example: HCl(g) ----> H+(aq) + Cl- (aq)
HCl is an acid because it increases the H+ concentration
Base:
- Any compound that increases hydroxide (OH-) concentration
Example: NaOH(s) ----> Na+(aq) + OH-(aq)
NaOH is a base because it increases the OH- concentration
Brønsted-Lowry Acids & Bases
** A more developed idea of Arrhenius acid/base concept
Acid:
- Proton donor (H+)
Base:
- Proton Acceptor
Acid:
- Proton donor (H+)
Base:
- Proton Acceptor
Lewis Acids & Bases
Acid:
- Electron pair acceptor
Base:
- Electron Pair donor
- Electron pair acceptor
Base:
- Electron Pair donor
Strong vs. Weak
AcidsA STRONG ACID will completely ionize (break apart into ions) in water all the time.
A WEAK ACID will only partially ionize some of the time MEMORIZE THESE STRONG ACIDS (all the rest are weak): - HCl -HBr -HI -H2SO4 - HNO3 - HClO4 |
BasesA STRONG BASE will completely ionize (break apart into ions) in water all the time.
A WEAK BASE will only partially ionize some of the time MEMORIZE THESE STRONG BASES (all the rest are weak): - ANY ELEMENT from the Alkali or Alkaline Earth Metals paired with an OH-. |
kwkw= [H+][OH-]
The kw of water will always be 1.00x10^-14 M If you add more H+ or OH-, the solution will balance out to 1.00x10^-14 |
Acidic, Basic, or Neutral?[H+] > [OH-] = Acidic
[H+] = [OH-] = Neutral [H+] < [OH-] = Basic |
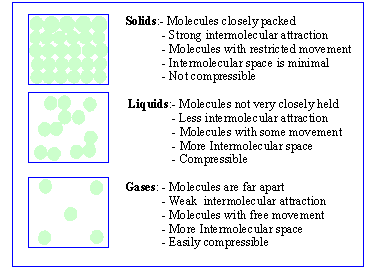
Intramolecular Forces
* Ionic Bonds
* Covalent Bonds
* Covalent Bonds
Intermolecular Forces
1. Dipole-Dipole- attraction between polar molecules with a partial positive and partial negative charge
2. Hydrogen Bonding- specific type of dipole-dipole that pertains to solely H and O
3. London Dispersion- holds nonpolar molecules together after their electrons randomly gather and cause the molecules to have partial positive or partial negative charges
2. Hydrogen Bonding- specific type of dipole-dipole that pertains to solely H and O
3. London Dispersion- holds nonpolar molecules together after their electrons randomly gather and cause the molecules to have partial positive or partial negative charges
Online Resources
pH Scale: http://chemwiki.ucdavis.edu/@api/deki/files/4116/=639px-PH_scale.png
Logarithms: http://www.wtamu.edu/academic/anns/mps/math/mathlab/col_algebra/col_alg_tut43_logfun.htm
Chapter 16 Review Quia (Good for math practice): http://www.quia.com/quiz/4220175.html
Sublimation and Deposition Slideshow: http://www.slideshare.net/weiheng1234/sublimation-and-deposition
Logarithms: http://www.wtamu.edu/academic/anns/mps/math/mathlab/col_algebra/col_alg_tut43_logfun.htm
Chapter 16 Review Quia (Good for math practice): http://www.quia.com/quiz/4220175.html
Sublimation and Deposition Slideshow: http://www.slideshare.net/weiheng1234/sublimation-and-deposition
Video Resources
How to Identify Acids, Bases, Conjugate Acids, and Conjugate Bases (by me!): https://vimeo.com/66112449
Phase Change Diagram Video: http://www.youtube.com/watch?v=2E4uK6zoJmo
Phase Change Diagram Video: http://www.youtube.com/watch?v=2E4uK6zoJmo
Activity Resources
pH Scale Quia Game: http://www.quia.com/rd/1975.html
Phase Change Jeopardy: http://www.superteachertools.com/jeopardyx/jeopardy-review-game-convert.php?gamefile=../jeopardy/usergames/Oct201144/jeopardy1320071436.txt
Phase Change Jeopardy: http://www.superteachertools.com/jeopardyx/jeopardy-review-game-convert.php?gamefile=../jeopardy/usergames/Oct201144/jeopardy1320071436.txt