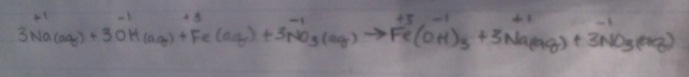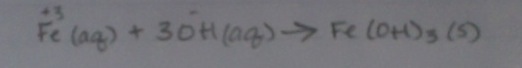Major Concepts• 4 Common "driving forces" for reactions (How can we tell if a reaction has occurred?)
• Solubility Rules (How can we know if a compound will be soluble?) • Predicting Products of Reactants in an Equation • Writing Complete Ionic Equations • Writing Net Ionic Equations • Types of Reactions 4 Common Driving Forces for Chemical Reactions
|
Example Problems1. Determine if this compound is soluble or insoluble... Fe(OH)2
Answer: (s), because most OH- compounds are insoluble. Fe(OH)2 is not an exception to the rule. 2. Determine if this compound is soluble or insoluble... (NH4)CO3 Answer: (aq), because even though it contains CO3, there is a rule that says compounds containing NH4 are soluble. 3. Determine the product: AgNO3(aq) + NaCl(aq) --> ____________ Answer: AgCl(s) + NaNO3(aq). Remember that AgCl is an exception to one of the solubility rules. Also note that reactants that are both soluble (aq) will have double displacement, where they switch partners. 4. Determine which type of chemical reaction has occurred:
Answer: Combustion. Remember, you need C and O in the reactants. Look for CO2 in the product! 5. Determine which type of chemical reaction has occurred:
Answer: Precipitation. Remember, look for two (aq) in the reactants and a solid in the product! |
Solubility Rules

Illustration of salt crystals dissolving in water. NaCl is a soluble compound.
1. Most nitrate (NO3-) salts are soluble.
2. Most salts of Na+, K+, and NH4+ are soluble.
3. Most chloride salts are soluble, except AgCl, PbCl2, and Hg2Cl2
4. Most sulfate salts are soluble, except BaSO4, PbSO4, and CaSO4
5. Most hydroxide compounds are insoluble, except NaOH, KOH, Ba(OH)2, and Ca(OH)2
6. Most sulfide, carbonate, and phosphate compounds are insoluble
Remember, if there are conflicting rules for a compound, USE THE RULE THAT DEEMS THE COMPOUND SOLUBLE (aq)!
2. Most salts of Na+, K+, and NH4+ are soluble.
3. Most chloride salts are soluble, except AgCl, PbCl2, and Hg2Cl2
4. Most sulfate salts are soluble, except BaSO4, PbSO4, and CaSO4
5. Most hydroxide compounds are insoluble, except NaOH, KOH, Ba(OH)2, and Ca(OH)2
6. Most sulfide, carbonate, and phosphate compounds are insoluble
Remember, if there are conflicting rules for a compound, USE THE RULE THAT DEEMS THE COMPOUND SOLUBLE (aq)!
Types of Equations (and how to write them!)1. Molecular Equation- this is the type of equation that we've been working with all year. It looks like a "normal" chemical equation to us. Here's our example:
3NaOH(aq) + Fe(NO3)3(aq) --> 3NaNO3(aq) + Fe(OH)3(s) 2. Complete Ionic Equation- this equation takes all aqueous solutions and separates them, symbolizing how aqueous solutions dissolved in water break apart. Leave solid compounds together. In this type of equation, we have to put the charges above each element. I'll use the same equation, just in complete ionic form now: 3. Net Ionic Equation- this equation concentrates on the solid in the product and the reactants that create the solid in. In this type of equation, we must also put the charges above each element. I'll use the same equation, just in complete ionic form now:
|
Types of Reactions1. Combustion- CH4+O2 --> CO2+H2O
In a combustion reaction, you will need to see carbon and oxygen in the reactants and carbon dioxide in the product. 2. Synthesis- H2+O2 --> H2O In a synthesis reaction, elements/compounds will combine to form a new single compound. 3. Decomposition- H2O --> H2+O2 In a decomposition reaction, a compound will break into simpler parts 4. Single Displacement- Zn+FeCl2 --> Fe+ZnCl2 In single displacement, one element or compound replaces another one in the equation. However, note that only a cation can replace a cation and only an anion can replace an anion. 5. Double Displacement- AgNO3(aq)+NaCl(aq) --> AgCl(s)+NaNO3 In double displacement, elements or compounds switch partners. Note that this reaction is also classified as a precipitation reaction (two aqueous compounds create a solid in the product) 6. Acid/Base- HNO3+NaOH --> H2O+NaNO3 In an acid/base reaction, an acid (H) and base (OH) react. Look for H2O in the product. An acid and a base will ALWAYS produce water! 7. Precipitation- AgNo3(aq)+NaCl(aq) --> AgCl(s)+NaNO3(aq) In a precipitation reaction, two aqueous compounds will form a solid in the product. Note that this reaction also has double displacement, where both of the compounds switch partners. |
Online Resources
Complete Ionic and Net Ionic Equations: http://www.occc.edu/kmbailey/chem1115tutorials/Net_Ionic_Eqns.htm
Types of Reactions (an explanation): http://misterguch.brinkster.net/6typesofchemicalrxn.html
Types of Reactions (an explanation): http://misterguch.brinkster.net/6typesofchemicalrxn.html
Video Resources
Soluble or Insoluble?: http://www.youtube.com/watch?v=I0xSc4DfTFk
Types of Equations (By me!): https://vimeo.com/54900253
Types of Equations (By me!): https://vimeo.com/54900253
Activity Resources
Classifying Types of Reactions Worksheet (With Answers!): http://misterguch.brinkster.net/6typesofreaction.pdf
Soluble or Insoluble Worksheet (With Answers!): http://chemistry.bd.psu.edu/halmi/wks4b1.pdfIf you can't get the .pdf to come up, save linked file as download and go to your downloads. It should be there!
Soluble or Insoluble Worksheet (With Answers!): http://chemistry.bd.psu.edu/halmi/wks4b1.pdfIf you can't get the .pdf to come up, save linked file as download and go to your downloads. It should be there!